
Differences in signal duration have been implicated as the basis of differential decisions in myriad cell processes. the time over which a kinase remains active). One key factor in specifying a cellular decision is the duration of a signal (i.e. For instance, although scaffolds are believed to have profound effects on the dynamics of signal propagation, ,, , the mechanisms that underlie how scaffolds regulate signaling dynamics are not well understood. Despite numerous advances in the understanding of the signaling function of scaffold proteins –, many questions remain. Signaling complexes maintained by scaffolds are intensely studied and have been shown to affect myriad cell decisions –. Signal propagation through these kinase cascades is often guided by a scaffolding protein that assembles protein kinases into a multi-protein complex. 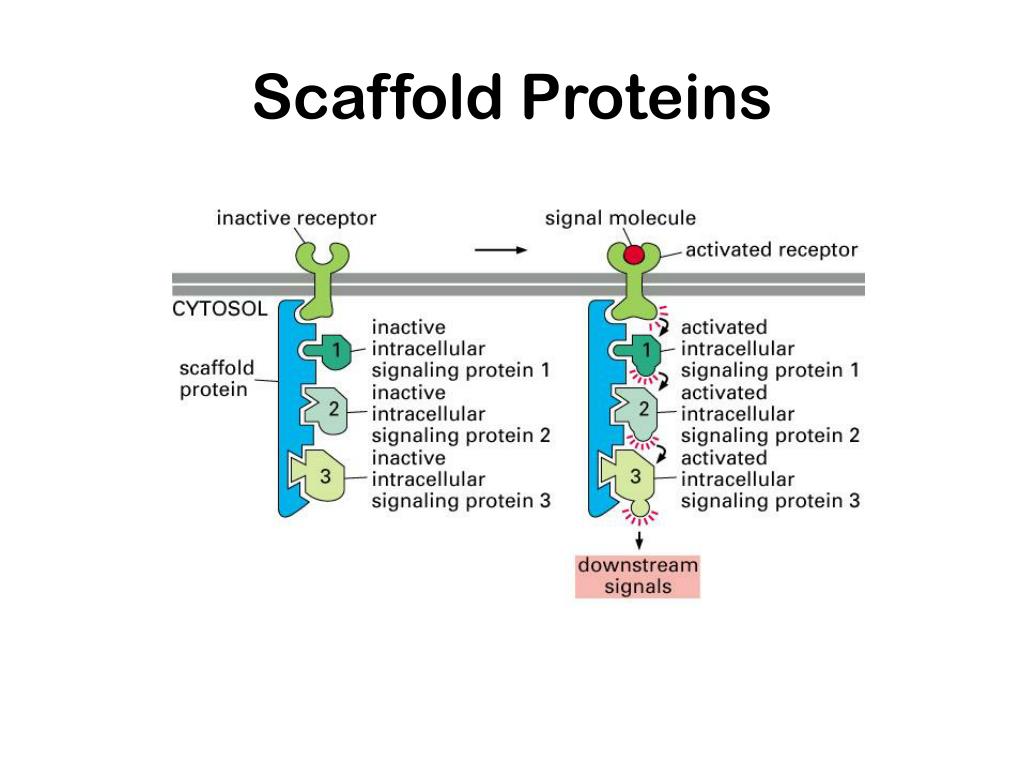
Within the complex networks that integrate these signals lies a highly conserved motif involving the sequential activation of multiple protein kinases. In the context of signal transduction, cells integrate signals derived from membrane proximal events and convert them into the appropriate cell decision. Our findings support the possibility that these signaling dynamics regulated by scaffolds affect cell decision-making in many medically important intracellular processes. Scaffolds allow for controlled levels of signal that are delivered inside the cell at appropriate times. 
The simulations reveal mechanisms for how scaffolds can dynamically regulate the timing of cell signaling. Therefore, we used a computational approach that simulates the behavior of a model signal transduction module comprising a set of proteins in the presence of a scaffold. Although scaffolds are widely believed to have dramatic effects on how signal transduction is carried out, the mechanisms that underlie these consequences are not well understood. Scaffolding proteins direct the assembly of multiple proteins involved in cell signaling by providing a platform for these proteins to carry out efficient signal transmission. For example, in higher organisms such as mammals, signal transduction that leads to cell proliferation is often guided by a scaffold protein. Tight regulation of signal transduction is required for all healthy cells, and aberrant signaling leads to countless diseases such as cancer and diabetes. Cells detect signals from their environment and use them to make decisions such as whether or when to proliferate. Signal transduction is the science of cellular communication. Our results illustrate another complexity in the broad array of control properties that emerge from the physical effects of spatially localizing components of kinase cascades on scaffold proteins. These calculations provide insights into one mechanism that describes how the duration of a signal can potentially be regulated in a scaffold mediated protein kinase cascade. Scaffold concentrations that result in optimal signal amplitude also result in the broadest distributions of times over which kinases are activated. Scaffolds can influence the timing of kinase activation by allowing for kinases to become activated over a broad range of times, thus allowing for signaling at both early and late times. The simulations indicate that a major effect that scaffolds exert on the dynamics of cell signaling is to control how the activation of protein kinases is distributed over time. Specifically, we examine the effects of spatially localizing kinase components on a scaffold on signaling dynamics. Here, Monte Carlo simulations of a model kinase cascade are used to investigate how the temporal characteristics of signaling cascades can be influenced by the presence of scaffold proteins. Although scaffolds are widely believed to have dramatic effects on the dynamics of signal propagation, the mechanisms that underlie these consequences are not well understood. 
Scaffolding proteins that direct the assembly of multiple kinases into a spatially localized signaling complex are often essential for the maintenance of an appropriate biological response.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
